
Cardiac Troponin I (cTnI) Test Kit | fluorecare® FIA POCT Manufacturer - Microprofit
Microprofit fluorecare® cTnI test kit is based on immunofluorescence assay (FIA) for quantitative detection of cardiac troponin I. Fast, accurate, and ideal for POCT and laboratory use.
- Model: MF-31
- Brand: fluorecare
- Specs: 20 tests/box,25 tests/box
Product Introduction
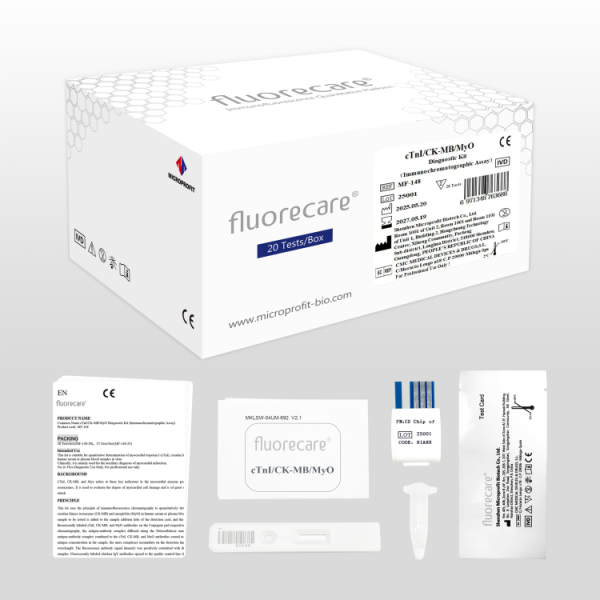
The fluorecare® cTnI Test Kit (Cardiac Troponin I) developed by Microprofit is a quantitative immunofluorescence assay (FIA) designed for the accurate detection of cTnI levels in human serum and plasma.
Cardiac troponin I is a highly specific biomarker for myocardial injury and is widely used in clinical diagnostics for the early detection and management of acute coronary syndromes.
As part of the fluorecare® POCT diagnostic platform, this test kit enables rapid and reliable quantitative analysis, supporting efficient clinical decision-making in emergency departments, laboratories, and point-of-care testing environments.
Detection Principle
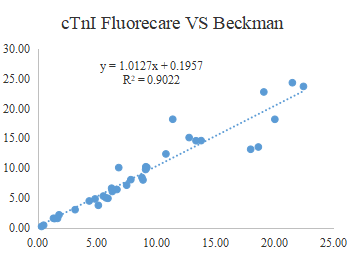
The fluorecare® cTnI test kit utilizes fluorescence immunochromatographic assay technology. cTnI in the sample binds with fluorescent-labeled antibodies to form antigen-antibody complexes, which are captured on the test line.
The fluorescence signal is detected and analyzed by the fluorecare® immunofluorescence analyzer, enabling accurate and quantitative measurement of cTnI concentration.
Clinical Applications
- Early detection and diagnosis of acute myocardial infarction (AMI)
- Risk stratification in patients with chest pain
- Monitoring of myocardial injury and cardiac events
- Evaluation of acute coronary syndromes (ACS)
- Clinical laboratory and emergency POCT testing
Frequently Asked Questions
What is the fluorecare® cTnI test kit used for?
The fluorecare® cTnI test kit is used for quantitative detection of cardiac troponin I to support diagnosis of myocardial injury and acute coronary syndromes.
How fast is the fluorecare® cTnI test?
The test provides accurate results within 15 minutes using the fluorecare® immunofluorescence analyzer.
What samples can be used for this test?
Serum and plasma samples are suitable for testing with the fluorecare® cTnI test kit.
Is the fluorecare® cTnI test suitable for POCT use?
Yes, it is specifically designed for point-of-care testing environments, especially in emergency and critical care settings.
Compatible Analyzer
The fluorecare® cTnI test kit is specifically designed to work with the fluorecare® immunofluorescence analyzer, providing fast, accurate, and quantitative results. This integrated POCT solution ensures efficient workflow and reliable performance for laboratories, clinics, and decentralized healthcare settings.
- Quantitative detection based on immunofluorescence assay (FIA)
- Rapid results within 15 minutes
- High sensitivity and specificity for cTnI detection
- Seamless integration with fluorecare® POCT platform
- Stable performance with internal calibration
- User-friendly operation with minimal training required
- Suitable for laboratory, emergency, and point-of-care testing scenarios
REF | MF-31 | |
Product Name | Cardiac Troponin I (cTnI) Diagnostic Kit (Immunochromatographic Assay) | |
Method | Immunochromatographic assay | |
Qualification | CE | |
Testing content | Cardiac Troponin I (cTnI) | |
Sample Type | Serum、plasma | |
Sample Volume(uL) | 70μL | |
Working Range | 0.1-50ng/ml | |
Reaction Time (min) | 15 | |
Storage Temperature | 2-30℃ | |
Shelf Life | 24 months | |
Applied Instrument | fluorecare® MF-T1000 Dry-type Immunofluorescence Quantitative Analyzer | |
Specification | 20 tests/kit | 25 tests/kit |
Size(L*W*H)mm/Kit | 145*115*75 | 205*138*83 |
Weight(kg)/Kit | 0.280 | 0.337 |
Kit components | Test Card:20 Cassettes ID Chip:1 Piece Instruction:1 Copy | Test Card ( including the desiccant):25 Cassettes ID Chip:1 Piece Instruction:1 Copy |